5 Key Takeaways
-
1
The SPECTRA trial evaluates the safety and efficacy of 4D-150 gene therapy for diabetic macular edema.
-
2
4D-150 is a novel intravitreal genetic medicine delivering a dual transgene cassette for aflibercept and VEGF-C inhibition.
-
3
Interim results show that a single dose of 4D-150 was well tolerated with no serious adverse events or intraocular inflammation.
-
4
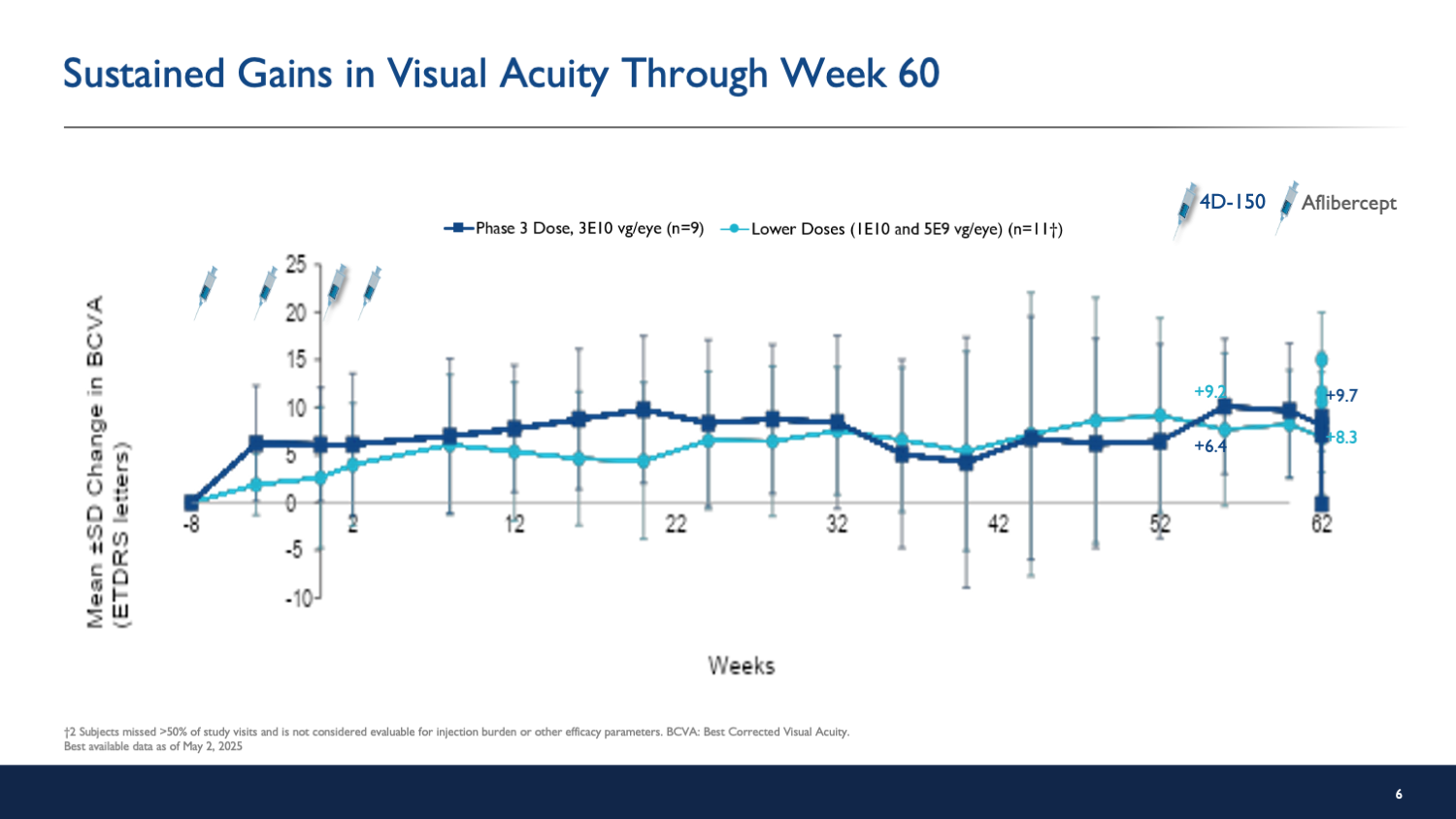
Patients in the high-dose group experienced an average gain of 8.4 letters and a 194 µm reduction in central subfield thickness.
-
5
The treatment significantly reduced the need for supplemental anti-VEGF injections by 84%, addressing patient concerns about treatment burden.
This content is an AI-generated, fully rewritten summary based on a published scholarly article. It does not reproduce the original text and is not a substitute for the original publication. Readers are encouraged to consult the source for full context, data, and methodology.