Objective:
To evaluate the safety and clinical activity of 4D-150 gene therapy for diabetic macular edema (DME), including its impact on treatment frequency.
Key Findings:
- 4D-150 was well tolerated with no serious adverse events or intraocular inflammation.
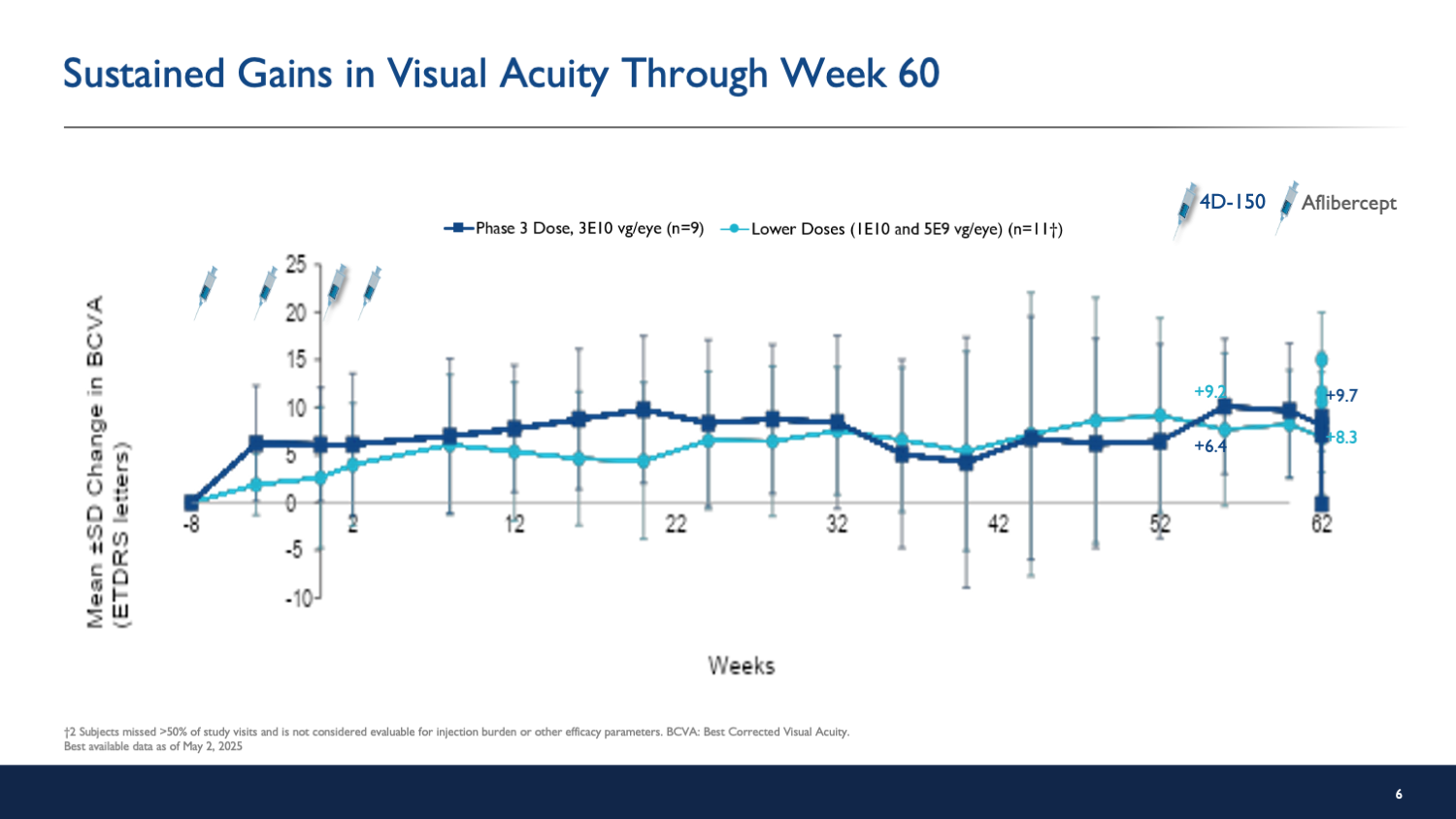
- Patients in the high-dose group gained an average of 8.4 letters in visual acuity, indicating significant improvement.
- Central subfield thickness reduced by 194 µm, showing anatomical improvement.
- There was an 84% reduction in treatment burden for the high-dose group, indicating a substantial decrease in the need for additional treatments.
Interpretation:
The interim results suggest that 4D-150 offers a promising safety profile and significant clinical benefits, potentially reducing the need for frequent anti-VEGF injections, which is a critical concern for patients.
Limitations:
- Interim data may not reflect long-term outcomes; further studies are needed to confirm durability and efficacy of 4D-150.
Conclusion:
A single injection of 4D-150 shows favorable safety and efficacy, significantly reducing treatment burden for DME patients.
This content is an AI-generated, fully rewritten summary based on a published scholarly article. It does not reproduce the original text and is not a substitute for the original publication. Readers are encouraged to consult the source for full context, data, and methodology.