Clinical Report: Dual-Action Gene Therapy for DME Shows Safety and Durability
Overview
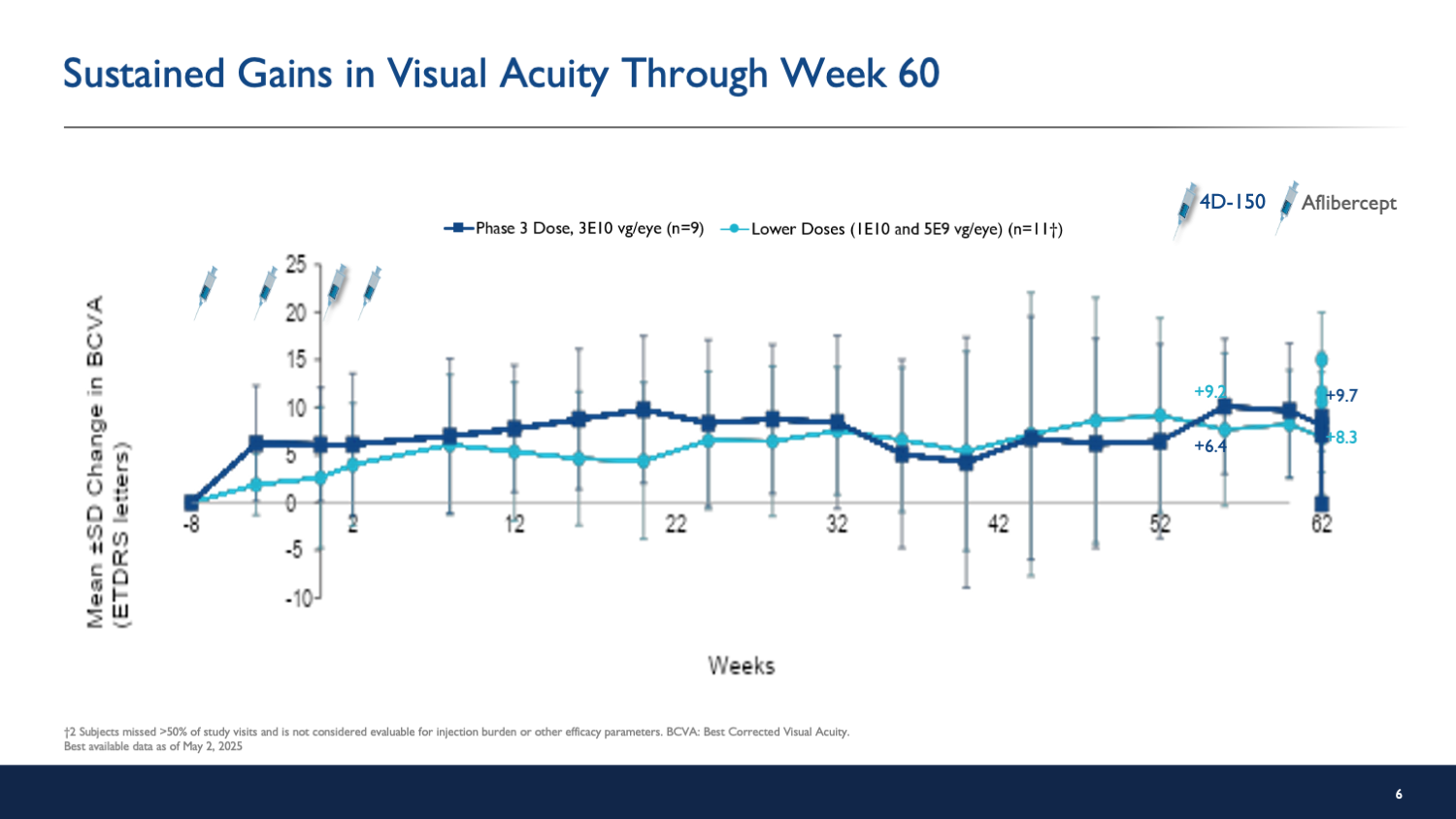
The SPECTRA trial presents interim results indicating that 4D-150 gene therapy is well tolerated and significantly reduces treatment burden in patients with diabetic macular edema (DME). A single injection of 4D-150 resulted in substantial visual and anatomical improvements without serious adverse events.
Background
Diabetic macular edema (DME) is a leading cause of vision loss in diabetic patients, necessitating effective and durable treatment options. Current first-line therapies, primarily anti-VEGF injections, require frequent administration, which can be burdensome for patients. The development of gene therapies like 4D-150 aims to provide longer-lasting effects and reduce the frequency of treatments.
Data Highlights
| Parameter | High-Dose Group |
|---|---|
| Average Letter Gain | 8.4 letters |
| Reduction in Central Subfield Thickness | 194 µm |
| Reduction in Treatment Burden | 84% |
Key Findings
- 4D-150 gene therapy demonstrated a favorable safety profile with no serious adverse events.
- Patients in the high-dose group experienced an average gain of 8.4 letters in visual acuity.
- There was a significant reduction in central subfield thickness by 194 µm.
- The treatment led to an 84% reduction in the need for supplemental anti-VEGF injections.
- 4D-150 is designed to deliver both aflibercept and a VEGF-C inhibitor, targeting multiple pathways in DME.
Clinical Implications
The results from the SPECTRA trial suggest that 4D-150 could represent a significant advancement in the treatment of DME, potentially reducing the treatment burden on patients while maintaining efficacy. Clinicians may consider this gene therapy as a viable option for patients seeking less frequent treatment regimens.
Conclusion
The interim results of the SPECTRA trial indicate that 4D-150 gene therapy is a promising approach for managing DME, offering both safety and significant reductions in treatment frequency. Further studies will be essential to confirm these findings and establish long-term outcomes.
References
- Retinal Physician, 2020 -- Adverum Gene-derived Anti-VEGF Proves Highly Durable
- Retinal Physician, 2024 -- The Landscape of Gene Therapy for Diabetic Retinopathy
- Retinal Physician, 2025 -- 4DMT SPECTRA trial interim results
- PubMed, 2025 -- Diabetic Retinopathy Preferred Practice Pattern®
- PMC, 2025 -- Pharmacological Treatment for Diabetic Macular Edema: A 2025 Update on Durability and Multi-Target Therapies
- Retinal Physician — Suprachoroidal Delivery as a Novel Avenue for Retinal Gene Therapy
- Diabetic Retinopathy Preferred Practice Pattern® - PubMed
- Pharmacological Treatment for Diabetic Macular Edema: A 2025 Update on Durability and Multi-Target Therapies - PMC
- 4DMT Presents Positive Interim Data from Intravitreal 4D-150 Phase 1/2 PRISM Clinical Trial in Patients with Wet AMD at ARVO 2023 | 4D Molecular Therapeutics
This content is an AI-generated, fully rewritten summary based on a published scholarly article. It does not reproduce the original text and is not a substitute for the original publication. Readers are encouraged to consult the source for full context, data, and methodology.