Clinical Scorecard: Dual-Action Gene Therapy for DME Shows Safety and Durability
At a Glance
| Category | Detail |
|---|---|
| Condition | Diabetic Macular Edema (DME) |
| Key Mechanisms | 4D-150 delivers a dual transgene cassette producing aflibercept and targeting VEGF-C. |
| Target Population | Adults with diabetic macular edema. |
| Care Setting | Multicenter clinical trial. |
Key Highlights
- 4D-150 showed a favorable safety profile with no serious adverse events.
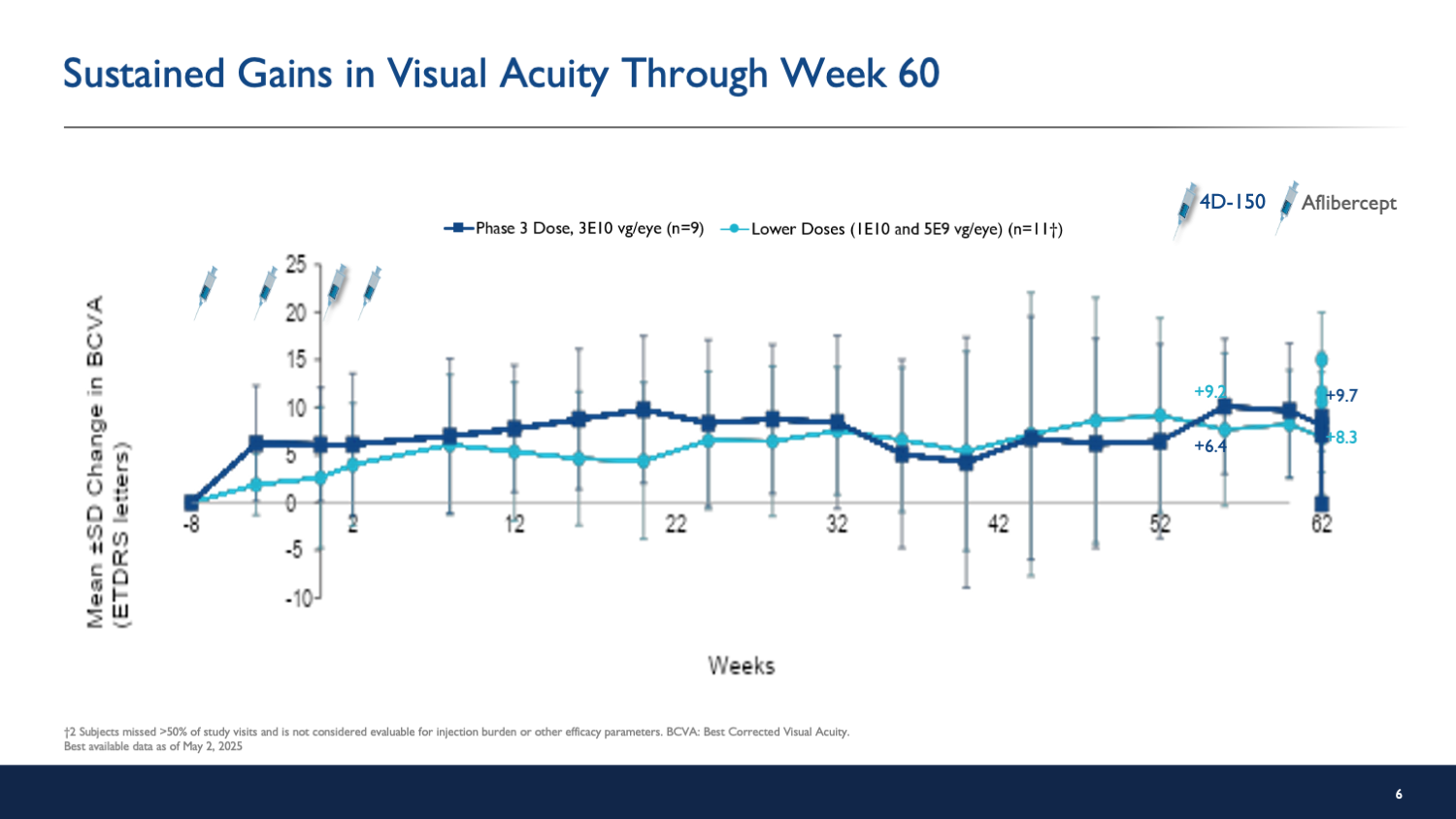
- Patients in the high-dose group gained an average of 8.4 letters in vision.
- Central subfield thickness reduced by 194 µm.
- 84% reduction in treatment burden for supplemental anti-VEGF injections.
- Single injection demonstrated sustained anatomical and visual gains.
Guideline-Based Recommendations
Diagnosis
- Evaluate patients with diabetic macular edema for potential gene therapy.
Management
- Consider initial aflibercept loading doses followed by 4D-150 injection.
Monitoring & Follow-up
- Monitor for safety and efficacy post-injection, particularly for intraocular inflammation.
Risks
- No reported serious adverse events related to 4D-150.
Patient & Prescribing Data
Adults with diabetic macular edema enrolled in the SPECTRA trial.
Single injection of 4D-150 significantly reduces treatment burden.
Clinical Best Practices
- Prioritize safety and efficacy in treatment planning for DME.
- Utilize gene therapy as a potential option for reducing treatment frequency.
References
This content is an AI-generated, fully rewritten summary based on a published scholarly article. It does not reproduce the original text and is not a substitute for the original publication. Readers are encouraged to consult the source for full context, data, and methodology.