Clinical Report: Complement Inhibition for Geographic Atrophy
Overview
Recent advancements in complement inhibition have led to FDA approval of pegcetacoplan and avacincaptad pegol for treating geographic atrophy (GA) secondary to age-related macular degeneration (AMD). These therapies represent a significant shift in managing GA, aiming to slow disease progression and improve patient outcomes.
Background
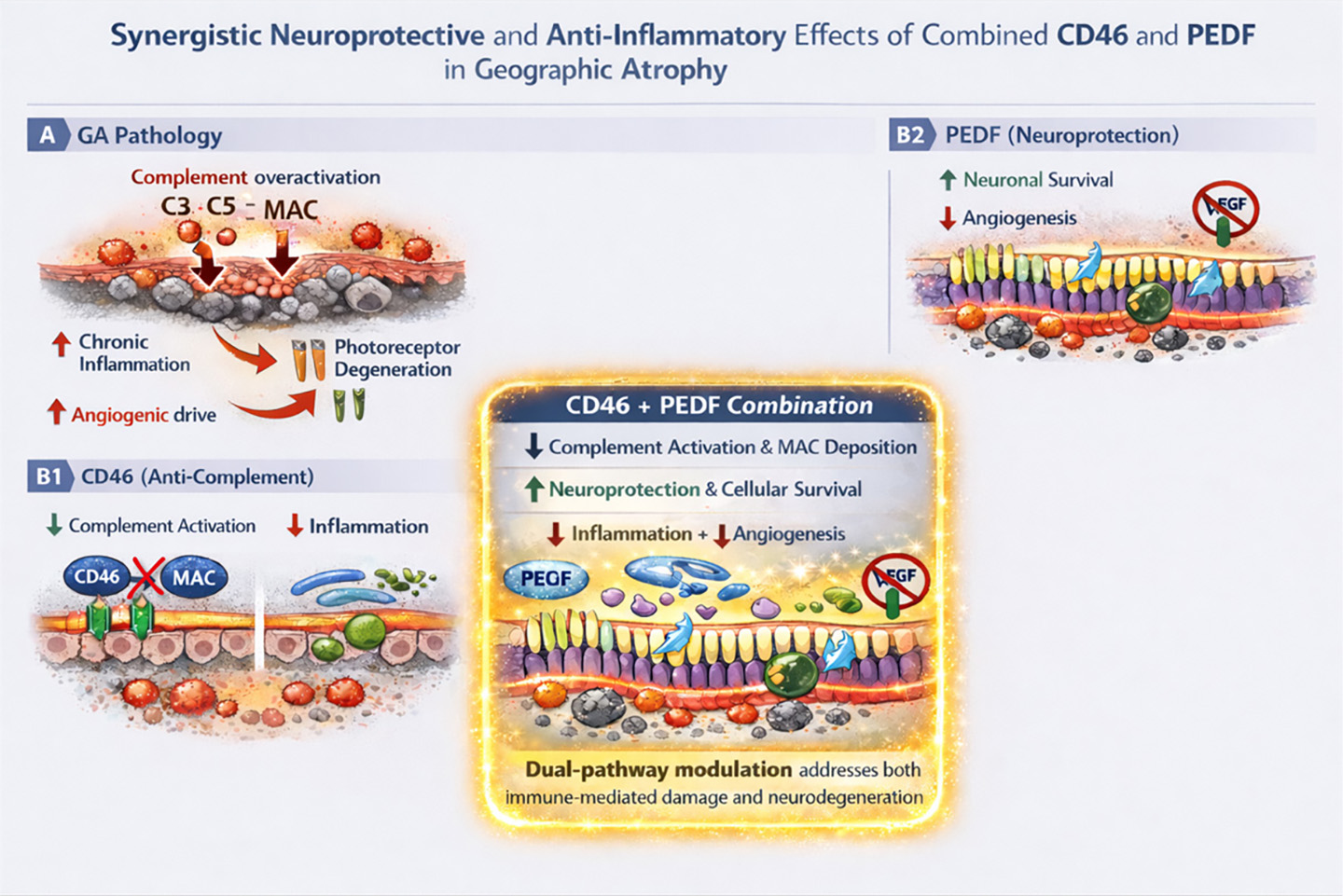
Geographic atrophy is a leading cause of irreversible vision loss in the elderly, linked to chronic low-grade inflammation and complement system dysregulation. The complement system plays a crucial role in immune response, and its inhibition has emerged as a promising therapeutic strategy to address GA. Understanding the mechanisms and implications of complement inhibition is vital for optimizing treatment approaches in AMD.
Data Highlights
No numerical data available in the source material.
Key Findings
- FDA approved pegcetacoplan and avacincaptad pegol as novel treatments for GA.
- Complement factor H mutations are implicated in over half of AMD cases.
- Elevated complement byproducts are found in retinal pigment epithelium and photoreceptor outer segments in GA-affected areas.
- Phase 3 trials OAKS and DERBY demonstrated significant slowing of GA lesion enlargement with pegcetacoplan.
- Avacincaptad pegol showed sustained slowing of GA growth without significant retinal vasculitis.
Clinical Implications
The introduction of complement inhibitors provides a new avenue for managing GA, shifting from watchful waiting to proactive treatment. Clinicians should consider the potential benefits and risks associated with these therapies, including the need for shared decision-making with patients regarding treatment options.
Conclusion
Complement inhibition represents a promising strategy in the management of geographic atrophy, with the potential to improve visual outcomes and quality of life for patients. Ongoing research and clinical experience will further elucidate the role of these therapies in AMD treatment.
References
- Retinal Physician, February 1, 2023 -- Treatment of Geographic Atrophy by Anatomic Location
- Retinal Physician, March-April 2026 -- Dual-Pathway Gene Therapy for Geographic Atrophy
- Ophthalmology Management, October 2025 -- Case Study: Preserving Vision in Geographic Atrophy with Complement Inhibitor Therapy
- Retinal Physician, July-August 2025 -- Complement Inhibition Slows GA in Eyes With Wet AMD
- AAO Preferred Practice Pattern - University of Miami -- Age-Related Macular Degeneration
- ScienceDirect -- Pegcetacoplan for the treatment of geographic atrophy secondary to age-related macular degeneration (OAKS and DERBY)
- PubMed -- Avacincaptad Pegol for Geographic Atrophy Secondary to Age-Related Macular Degeneration: Two-Year Efficacy and Safety Results from the GATHER2 Phase 3 Trial
- AAO Preferred Practice Pattern - University of Miami
- Pegcetacoplan for the treatment of geographic atrophy secondary to age-related macular degeneration (OAKS and DERBY): two multicentre, randomised, double-masked, sham-controlled, phase 3 trials - ScienceDirect
- Avacincaptad Pegol for Geographic Atrophy Secondary to Age-Related Macular Degeneration: Two-Year Efficacy and Safety Results from the GATHER2 Phase 3 Trial - PubMed
This content is an AI-generated, fully rewritten summary based on a published scholarly article. It does not reproduce the original text and is not a substitute for the original publication. Readers are encouraged to consult the source for full context, data, and methodology.