The development of anti-VEGF therapy has revolutionized the management of neovascular age-related macular degeneration (nAMD) and diabetic macular edema (DME), considerably improving the outlook for patients to maintain functional vision.
During the early years of anti-VEGF therapy, clinicians began to recognize that there is a relationship between fluctuations in retinal thickness and long-term visual acuity outcome. In clinical trials, investigators adhere to a strict treatment schedule. But in the real world, it became apparent that continuous monthly or even bi-monthly therapy with intravitreal injections was not a practical solution, due to multiple reasons, including poor patient compliance.1 Attempts were made to develop more pragmatic, less frequent treatment approaches, beginning with a PRN regimen, which the PrONTO study showed was an effective way to manage patients, provided patient visit tempo remained monthly.2
One of the lessons learned from PRN therapy is that withholding treatment until we detect evidence of recurrent fluid instigates episodes of fluid fluctuations, which in turn negatively affects post-treatment visual acuity. Our real-world observations were verified in clinical studies, demonstrating that fluid fluctuations matter.
CST Variability: A More Reliable Prognostic Marker
Some of the most compelling data linking fluid fluctuations with visual outcomes in nAMD was seen in a post-hoc analysis of the Comparison of Age-Related Macular Degeneration Treatments Trials (CATT) and the Inhibition of VEGF in Age-Related Choroidal Neovascularization (IVAN) trial.3 Both trials compared visual acuity outcomes with ranibizumab and bevacizumab given monthly or as needed in patients with neovascular AMD and divided patients into quartiles based on the magnitude of fluid fluctuation.
A post-hoc analysis showed that patients in the lowest quartile—those who exhibited the least amount of fluid changes, visit to visit—had significantly better visual outcomes than patients in the highest quartile. At 2 years, there was a substantial difference in visual acuity between the lowest quartile and the highest quartile, with a mean difference between them ranging from 6 to 9 letters. This analysis also showed that eyes with greater fluctuations in retinal thickness were more likely to develop fibrosis and geographic atrophy (Figure 1).
To minimize retinal fluid fluctuations and avoid the associated detrimental effects, the community switched to a treat-and-extend approach: treating at every visit unless the retina is dry, then trying to increase the interval between visits. Based on my personal clinical experiences and those of colleagues, I’ve observed that often, when treatment intervals were extended, fluid fluctuations would occur more frequently. In some of these cases, patients who experienced significant fluid fluctuations did not always regain vision.
Subsequent clinical trials confirmed our real-world observation that fluid fluctuations are associated with poorer visual acuity outcomes after anti-VEGF treatment.
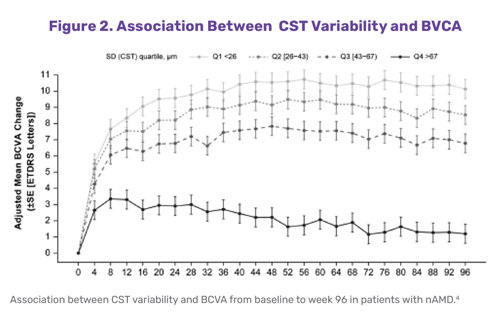
A post-hoc analysis of the HAWK and HARRIER trials of brolucizumab and aflibercept in neovascular AMD, which also divided patients into quartiles based on the magnitude or severity of fluid fluctuations, showed an 8-letter difference between the first and fourth quartiles.4 The researchers concluded that stable central subfield thickness (CST) was associated with better visual acuity outcomes after treatment (Figure 2). Their findings suggest that CST variability may be a more reliable prognostic marker of visual outcomes than the presence of fluid alone.
Another post-hoc analysis of the HAWK and HARRIER trials compared reductions in subretinal hyperreflective material (SHRM) thickness after treatment with brolucizumab versus aflibercept.5 Researchers found that greater fluctuations in SHRM led to substantially worse visual outcomes. In fact, many of the patients who had the most severe fluctuations in SHRM experienced a net decline in vision. Looking at individual cases, researchers also observed that, as the SHRM was allowed to come back, patients developed more persistent fibrosis.
When post-hoc analyses of multiple trials all demonstrate similar outcomes, the findings represent compelling data that warrants the attention of the retina community and should be factored into clinical decision-making.
Addressing the Challenges
Most retina specialists in the United States use a treat-and-extend strategy for their patients with AMD and DME. In an ideal world, treat-and-extend should do a relatively good job in reducing fluid fluctuations for most patients, but in the real world, some challenges persist.
First, in the context of a busy clinical practice, most retina specialists are faced with the challenge of rapidly reviewing and comparing OCT scans between visits, and typically just the prior visit. Although artificial intelligence–based tools to quantify fluid in specific compartments have been developed, they are not broadly available or used by most practitioners. Thus, clinicians may not fully appreciate the magnitude of fluid fluctuations that may be occurring in their patients, particularly in the longer-term course of treatment.
Second, AMD patients are elderly. They may have other health issues, and they often need caregivers to bring them to their appointments. However, caregivers may have scheduling conflicts, and as a result, patients miss visits. DME patients may have comorbid illnesses that require frequent medical follow up. This is a working-age population, so these people have jobs and other responsibilities that cause them to miss appointments.
So, how do we overcome these challenges? Better disease control is one potential answer.
Recently, faricimab, a novel bi-specific agent that targets both VEGF and angiopoietin-2, and aflibercept 8 mg, have become available and also appear to achieve greater durability of disease control. While these agents can last longer, we know that fluid fluctuations can still occur in this setting, especially with even longer dosing intervals. This is perhaps to be expected as the fluctuations are likely related to the pharmacokinetics of these agents in the eye, as they are given as a bolus injection with a sharp spike in drug concentration immediately after injection, followed by clearance from the eye which may not be linear. Thus, the intraocular drug concentration can be well above the therapeutic level required to suppress exudation immediately after the injection, but then fall well below this level before the next injection is given, likely contributing to fluctuations.
Another promising strategy that can help smooth out the curves in terms of fluid fluctuations involves a surgically implanted continuous drug delivery device. The Archway trial evaluated the Port Delivery System (PDS) with ranibizumab versus monthly ranibizumab intravitreal injections to treat neovascular AMD. In a post-hoc analysis, researchers found that eyes treated with the ranibizumab PDS maintained best-corrected visual acuity to 96 weeks regardless of retinal fluid or CST fluctuations, and this was comparable to monthly ranibizumab.6
In other words, continuous delivery of an anti-VEGF agent could achieve stable vision without fluid fluctuations, which potentially addresses the real-world challenges of missed appointments. A limitation of the ranibizumab PDS is that it requires surgical implantation of a device, which does carry some risks.
Sustained Release Tyrosine Kinase Inhibitors: A Promising New Alternative
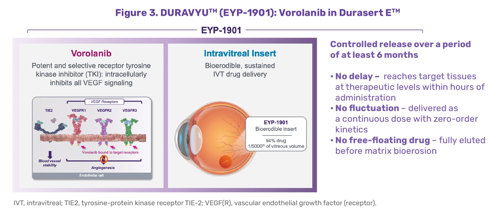
Alternative methods for continuous drug delivery involve in-office, intravitreal injections. One such potential therapy is DURAVYU™ (vorolanib intravitreal insert; also known as EYP-1901; EyePoint Pharmaceuticals). This investigational therapy combines vorolanib, a potent and selective tyrosine kinase inhibitor (TKI) that inhibits VEGF receptors 1, 2, and 3 and the PDGFR, with Durasert E™, a bioerodible insert (Figure 3). The result is a drug insert that is immediately bioavailable that continuously releases vorolanib for at least 6 months with no dose fluctuations.3-5
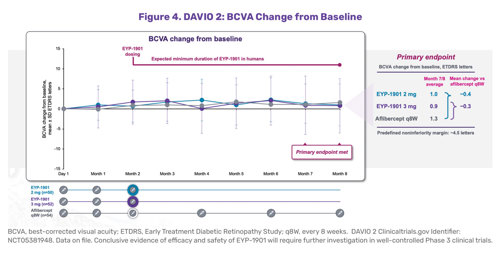
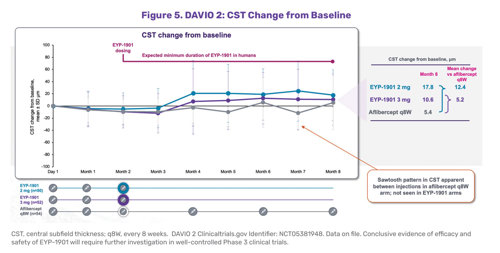
The Phase 2 DAVIO 2 clinical trial evaluated DURAVYU as a potential every 6-month drug to treat wet AMD.7 Two different doses of vorolanib were compared to aflibercept every 8 weeks and at weeks 28 and 32, the blended primary endpoint. DURAVYU-treated patients had nearly identical best-corrected visual acuity compared to patients treated with on-label aflibercept at the 24-week endpoint (Figure 4). DURAVYU-treated patients also showed strong anatomic control, with little to no fluctuations of the retina and had clinically meaningful, supplement-free rates (Figure 5).
Basically, this study showed that a single dose of the vorolanib intravitreal insert kept the retina dry for at least 6 months with stable vision and strong anatomical control on par with aflibercept, given every 8 weeks; however, there was one key difference – DURAVYU patients had minimal fluid fluctuations. If Phase 3 results confirm what was observed in Phase 2, this is very promising. Having a sustained-release intravitreal insert continuously treating the disease in the background without the need for surgery could potentially help address issues related to patients who might otherwise miss a visit and could hopefully reduce the number of retinal fluid fluctuations in real-world situations.
In Conclusion: Progress on Several Fronts
Looking to the future, other long-acting treatment strategies are in development—including gene therapy to transform retinal cells to continuously produce anti-VEGF molecules in the eye, as well as separate efforts involving other TKIs. These various emerging treatments offer great hope to our patients and suggest that real-world results may more closely mimic clinical trial results in the future.
Dr. Sadda is a paid consultant for EyePoint Pharmaceuticals; however, the statements made are the opinions of Dr. Sadda for educational purposes only. His statements are not intended as medical advice or the opinion of EyePoint.

References
- Khurana RN, Li C, Lum F. Loss to follow-up in patients with neovascular age-related macular degeneration treated with anti-VEGF therapy in the United States in the IRIS® Registry. Ophthalmology. 2023;130(7):672–683. doi:10.1016/j.ophtha.2023.02.0211
- Lalwani GA, Rosenfeld PJ, Fung AE, et al. A variable-dosing regimen with intravitreal ranibizumab for neovascular age-related macular degeneration: Year 2 of the PrONTO Study. Am J Ophthalmol. 2009;148(1):43–58. doi:10.1016/j.ajo.2009.01.024
- Evans RN, Reeves BC, Maguire MG, et al. Associations of variation in retinal thickness with visual acuity and anatomic outcomes in eyes with neovascular age-related macular degeneration lesions treated with anti-vascular endothelial growth factor agents. JAMA Ophthalmol. 2020;138(10):1043–1051. doi:10.1001/jamaophthalmol.2020.3001
- Dugel PU, Jhaveri CD, Chakravarthy U, et al. Effect of retinal thickness variability on visual outcomes and fluid persistence in neovascular age-related macular degeneration: A post hoc analysis of the HAWK and HARRIER studies. Retina. 2022;42(3):511–518. doi:10.1097/IAE.0000000000003349
- Sadda S, Sarraf D, Khanani AM, et al. Comparative assessment of subretinal hyper-reflective material in patients treated with brolucizumab versus aflibercept in HAWK and HARRIER. Br J Ophthalmol. 2024;108(6):852–858. doi:10.1136/bjo-2023-323577w
- Sheth VS, Holekamp NM, Khanani AM, et al. Retinal fluid and thickness fluctuations in Archway trial for port delivery system with ranibizumab versus monthly ranibizumab injections. Ophthalmol Retina. 2025;9(4):330–342. doi:10.1016/j.oret.2024.10.015
- EyePoint pharmaceuticals announces positive topline data from the Phase 2 DAVIO 2 Trial of EYP-1901 in wet AMD achieving all primary and secondary endpoints. Press release. EyePoint Pharmaceuticals. December 4, 2023. Accessed June 2, 2025. https://investors.eyepointpharma.com/news-releases/news-release-details/eyepoint-pharmaceuticals-announces-positive-topline-data-phase-2
Sponsored by ![]()










