The movement of immune cells through the retinal vasculature has long been inferred from laboratory models. Now, advances in imaging are allowing clinicians to observe these processes directly. At the 2026 meeting of the Association for Research in Vision and Ophthalmology (ARVO) in Denver, Richard B. Rosen, MD, ScD(hon), FACS, FASRS, FARVO, described early clinical work using adaptive optics scanning laser ophthalmoscopy (AOSLO) to visualize leukocyte trafficking in vivo.
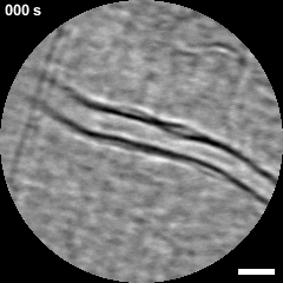
Leukocytes play a central role in immune surveillance, moving along the vascular endothelium through a sequence of rolling, crawling, adhesion, and transmigration. “Leukocytes patrol the vascular highways, really providing local immune surveillance,” explained Dr. Rosen, surgeon director and retinal service chief at the Mount Sinai Health System and vice chair of ophthalmology research at the Icahn School of Medicine at Mount Sinai. “Leukocyte crawling along the vascular surface is part of the cascade, which actually starts with rolling, and then the cells slow down to crawl, and then gradually progress to adhesion.”
Until recently, these dynamics were largely confined to in vitro systems or animal models. AOSLO enables high-resolution imaging of retinal microvasculature by correcting for optical aberrations and capturing rapid sequences that can be analyzed over time. In this study, investigators combined optical coherence tomography angiography (OCTA) to identify regions of interest with high-speed video acquisition to track cell movement. The work builds on prior experimental imaging but adapts it for clinical use with a commercially available system from Robotrak. “Clinical commercial AOSLO is now consistently able to visualize leukocyte crawling noninvasively, both in healthy and diseased eyes,” he said.
Dr. Rosen presented data from a small cohort of healthy participants (n=16) who served as controls and patients with diabetic retinopathy (n=5), sickle cell disease (n=2), and uveitis (n=5). Investigators acquired short video sequences and manually tracked leukocyte motion. Distinct patterns were observed, including movement with and against flow, backtracking, and lateral migration across vessel walls. Leukocyte activity was more frequently observed in venules than arterioles, with an approximate 10:1 difference.
Cell behavior appeared consistent across vessel sizes, with measured velocities showing limited variation. “There wasn’t a big difference in the speed of these cells, depending on the size of the lumen,” Dr. Rosen said. “They seem to have their own particular velocity.”
Case examples illustrated how these dynamics may vary in disease. In diabetic retinopathy, leukocyte stalling and adhesion were observed near areas of nonperfusion and microaneurysms. In sickle cell disease, both adhesion and occlusive behavior were noted, along with evidence of transmigration. In uveitis, imaging suggested increased extravascular activity, with cells visible both within and outside vessel walls.
Dr. Rosen also described a case in which leukocyte activity appeared to change following anti-VEGF therapy. “When the patient came back a month later, after having anti-VEGF, in the exact same area, these cells seemed to be moving much more,” he said, noting that the significance of this observation remains unclear.
The ability to visualize leukocyte dynamics in vivo may provide insight into disease mechanisms and treatment effects. “The evaluation and characterization of these activities offer a dynamic window of the immune system, which may impact our understanding of disease and inform our therapies,” Dr. Rosen said. RP








